Road Traffic Signalling Batteries
Road traffic signalling zinc air-alkaline batteries are EXTREME SAFETY. Long operating time. High and constant voltage.Flashing lights, Cascades.
ZINC AIR BATTERIES use atmospheric oxygen to produce electrochemical energy. Since one active material lies outside of the cell, the majority of the cell‘s volume contains the other actice component (zinc), so zinc air batteries have very high energy density. Using a reactant from the air saves on space, reducing the size and weight of the battery.
ZINC AIR-ALKALINE BATTERY
Higher discharge current (50mA or 150Ω).
Higher and constant voltage (8V).
Higher energy density (350Wh/kg).
Operating time: 2 years.
ZINC AIR-SALINE BATTERY
Lower discharge current (25mA or 300Ω).
Average voltage: 6V.
Energy density (150Wh/kg).
Operating time: 3 years (lower shelf-discharging).
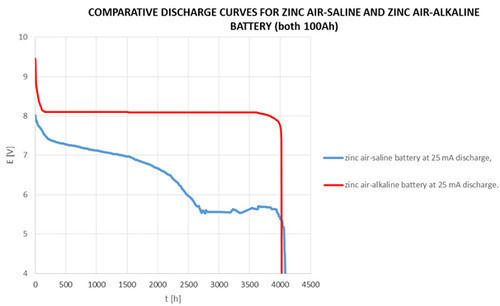
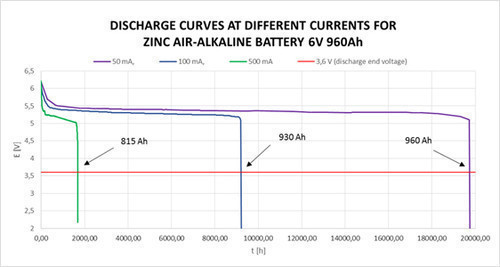
ZINC AIR-ALKALINE BATTERIES are based on a zinc-oxygen electrochemical couple. They use atmospheric oxygen (cathode) for producing depolarization. Anode is the large surface area zinc powder disperzed in a potassium hydroxide solution.
- Higher energy density and specific energy ratio than other types of batteries: 350 Wh/kg.
- The best energy cost due to the high energy density.
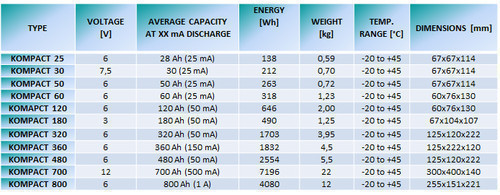
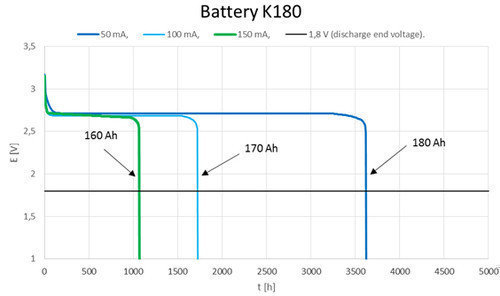
- High and constant output voltage (1,5V; 3V; 4,5V; 6V; 7,5V; 9V; 12V; 15V).
- Require no maintenance and no special procedures before being ready to use.
- Activated by removing label or stopper on the top of the battery.
- Safe to handle, without any risk of leakage on fixed or moving installations.
- Operating time 2 years (capacity loss for each year is just 5%).
- Can be satisfactorily used at temperatures ranging from -20°C to +45°C.
- Not subject to any special transport condition.
ZINC AIR BATTERIES are best suited to applications where the devices demand continuous low power load, isolated from the network (for a medium to long-term period of time).
-
Contact Sales
Jože RUS
E-mail Contact card -
Technical Documentation
-
Certificates
